ASH Career Development Awards
As the world's largest professional society of hematologists, ASH is dedicated to supporting hematologists in all career stages. Through more than 20 ASH awards, medical students, residents, and hematologists in all sub-specialty areas can find opportunities to receive mentorship and funding, complete a research project, gain recognition from peers, and ultimately advance their careers in the dynamic field of hematology. ASH Awards also honor those who have helped advance the field of hematology in significant ways.
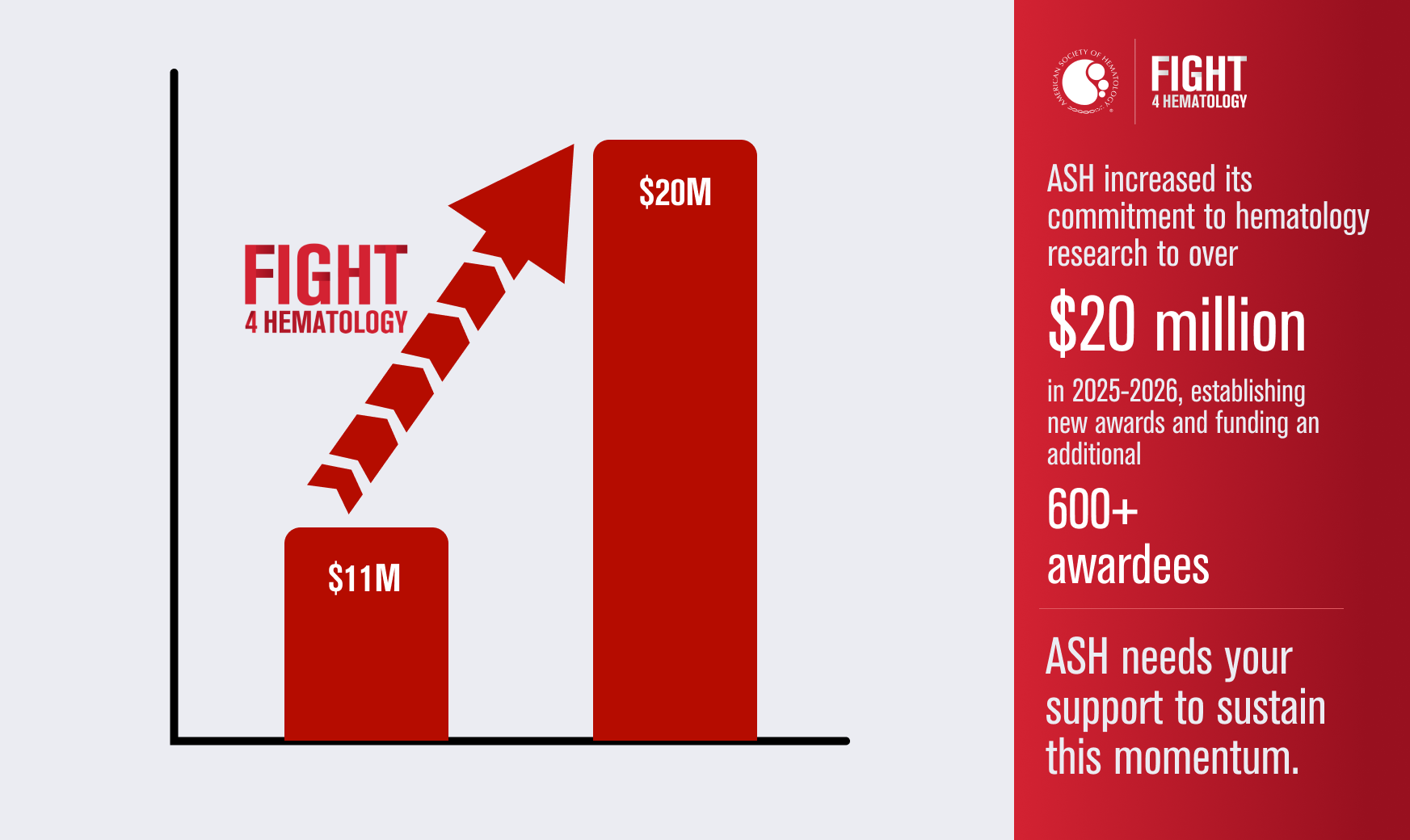
Join ASH to Help Bridge Funding Gaps
ASH increased its commitment to funding critical hematology research this year by $9 million to establish new awards and fund an additional 600+ awards to make up for the gaps in opportunity resulting from federal funding cuts – bringing ASH’s total investment to more than $20 million in 2025-2026. It’s going to take your help to sustain that momentum beyond this first year. Donate to the ASH Foundation to help protect the future of hematology research when it’s needed most.
ASH Awards for All Career Stages
Medical Student, Resident, Graduate Student Awards
ASH offers both short- and long-term opportunities for aspiring hematologists in medical school or residency to conduct hematology research under the supervision and mentorship of ASH member mentors. Apply for the chance to conduct hands-on research and jump-start your career.
Early and Mid-Career Investigator Awards and Training Programs
ASH offers multiple funding and training opportunities for hematology fellows, junior faculty, and senior research faculty at various career stages. Apply for one of our career-enhancement awards or training programs to get the research funding, training, or guidance needed to move your hematology career forward.
ASH Abstract Achievement Awards and Outstanding Abstract Achievement Awards
Each year, ASH offers a variety of merit-based awards to support select trainees with high-achieving annual meeting abstracts who are chosen to present their work at the ASH annual meeting.
ASH Honorific Awards
ASH's most prestigious awards, the Honorific Awards recognize hematologists who have made significant contributions to the field of hematology.
Hematology Inclusion Pathway
The ASH Hematology Inclusion Pathway (HIP), the successor program to the ASH Minority Recruitment Initiative (MRI), offers a comprehensive 13-year longitudinal career pathway of awards that strives to ensure that hematology reflects and serves the world we live in– from medical students to mid-career professionals.
The HIP Initiative aligns with ASH’s commitment to excellence through expansion and inclusion and reflects an evolving understanding of the hematology community.
With HIP, we’re continuing to expand the availability of hematologic care while promoting the highest standards of excellence in hematology research, training, and practice.



